
Reckitt unit hit with $60 million verdict in Enfamil baby formula case in Illinois Reckitt shares sink as investors see more trouble from baby formula lawsuits US FDA approves Abbott's heart valve repair deviceĪbbott Receives FDA Approval for Triclip?, First-Of-Its-Kind Device to Repair Leaky Tricuspid Heart ValveĪbbott Laboratories Says Its i-STAT TBI Cartridge Received US FDA Clearance for Use With Whole BloodĪbbott Laboratories Cartridge Gets FDA Clearance to be Used with Whole BloodĪbbott Receives FDA Clearance for Whole Blood Rapid Test to Help with Assessment of Concussion at the Patient's BedsideĪbbott Laboratories' Assert-IQ Insertable Cardiac Monitor Gets CE Mark in EuropeĪbbott Advances Chronic Pain Management with Small Implanted, Rechargeable Spinal Cord Stimulation System S&P 500 Posts Second Consecutive Weekly Drop Led by Financials Amid Higher-Than-Expected Consumer PricesĬitigroup Raises Price Target on Abbott Laboratories to $128 From $126, Keeps Buy RatingĪbbott's heart valve repair device gets US approvalĪbbott Laboratories' TriClip Receives FDA Approval for Tricuspid Valve RepairĪbbott Gets FDA Approval for TriClip Heart Valve Repair System The Eterna SCS System is currently the small device available, designed for implant site comfort, and features Abbott's BurstDR stimulation, which mimics natural firing patterns found in the brain to deliver superior pain relief over traditional tonic stimulation. The studies found that people wanted small, comfortable implants which don't need to be charged daily.Ībbott designed the Eterna SCS System to be recharged wirelessly at a frequency of five times per year under normal use, making it the lowest recharge burden platform on the market. Approved by Health Canada in 2023, Abbott developed the Eterna SCS System based on extensive studies with patients, physicians, and caregivers to understand the unmet needs of people living with chronic pain. The Eterna SCS system uses Abbott's proprietary BurstDR stimulation, the only SCS technology with the high level of clinical evidence (level 1A.) Abbott's proprietary BurstDR therapy is proven to reduce pain by 23% more than traditional tonic stimulation without the tingling sensation. The inaugural use case of the Eterna SCS system took place at Hôpital de l'Enfant-Jésus hospital in Quebec City. See product advisories.Abbott announced that the company's Eterna SCS system ? the small implantable, rechargeable spinal cord stimulator (SCS) for the treatment of chronic pain, has been used for the first time in Canada. The inability to exit MRI mode would require device replacement surgery to restore therapy. Note: For Proclaim™ family of devices, performing any of the above actions while your device is in MRI mode may result in the inability to exit MRI mode. DO NOT: update, install, or delete the Patient Controller app or Bluetooth ‡ connection.DO NOT: alter, damage, update or lose your Patient Controller handheld.Set your system to MRI mode. This turns off stimulation and protects your device during your MRI scan.If applicable, fully charge your generator. 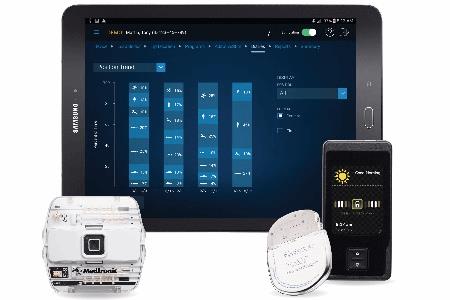 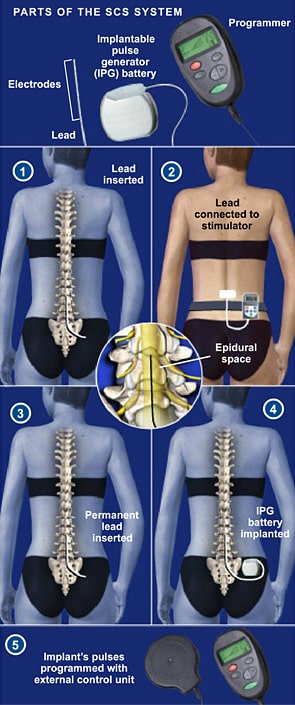
Refer to your user guide for charging instructions. 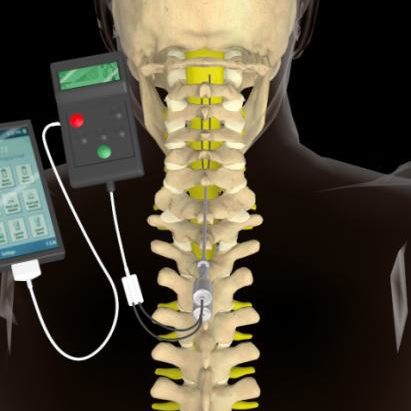
Disable automatic updates for the Patient Controller app and iOS ‡ software before placing the IPG in MRI mode.For Proclaim™ family of devices, before attempting to place your device into MRI mode, and with the help of your physician or Abbott representative, ensure your Patient Controller app is updated to the latest “ Patient Controller NR - US” application from the Apple ‡ App Store ‡.Please see guidance currently available in your Patient Manual. The Abbott devices shown in the table are eligible for an MRI scan within approved parameters.Īlways ask your pain physician to confirm whether your device is eligible for an MRI.ĭepending on your device and which leads have been implanted with your system, you may qualify for an MRI scan of either your whole body or just your head and extremities (arms and legs). Your system may be designed to let you safely receive an MRI scan under certain conditions. Our table can help you identify which kinds of scans are approved for each of our MR Conditional implanted devices. If you have an implanted device, you need to make sure your device is approved to receive an MRI.įortunately, with certain Abbott neurostimulators, you can safely have an MRI scan when conditions for safe scanning are met. But in rare cases, they can interfere with your neurostimulator device, possibly leading to injury. In general, MRI scans are safe, routine procedures. 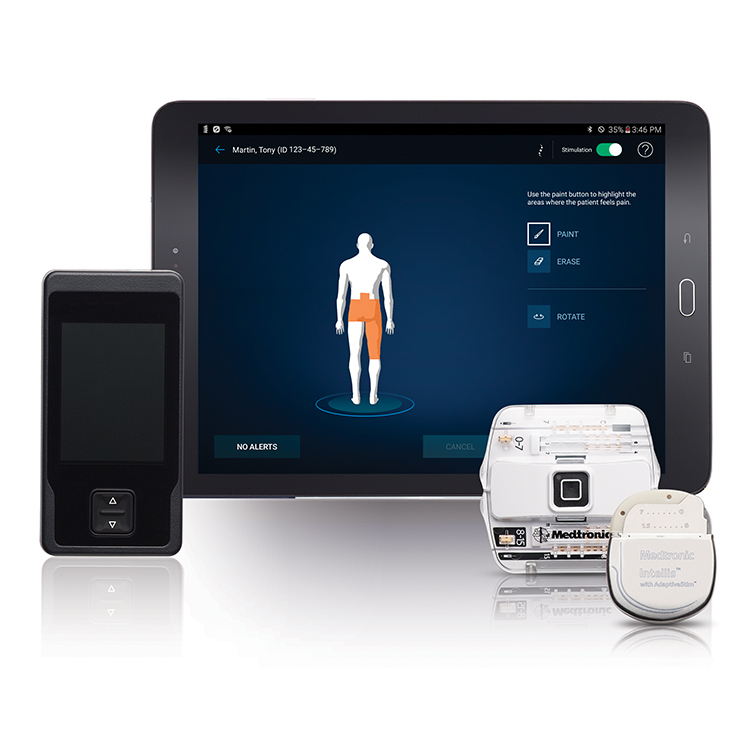
An MRI uses strong magnets to create images of the organs and tissues in the body. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
